particle in a box approximation of metals In quantum mechanics, the particle in a box model (also known as the infinite potential well or the infinite square well) describes the movement of a free particle in a small space surrounded by impenetrable barriers. The model is mainly used as a hypothetical example to illustrate the differences between classical and quantum systems. In classical systems, for example, a particle trapped inside a large box can move at any speed within the box and it is no more likely to be fo. What else the extremely compact and inexpensive loudspeaker has to offer and in particular, how it can hold its own against my ultimate budget favourite, the Xiaomi Mi Square Box Bluetooth Speaker, you will find out in my following review.
0 · particle in a box wikipedia
1 · particle in a box model
2 · particle in a box function
3 · particle in a box equation
4 · particle in a box diagram
5 · particle in a box
6 · formula for particle in a box
7 · 2 dimensional box particle formula
American-made, architectural-grade woven wire mesh for custom ceiling treatments, fixed or operable metal partitions/dividers, pre-engineered security gates/screens, metal wall coverings, metal mesh drapery, curtains and window treatments, and a lot more, sustainably manufactured to specification only by Cascade Architectural.
The particle-in-a-box eigenfunctions are given by Equation \(\ref{3.5.14}\), with \(B = 0\) and \(k = n\pi/L=a\), in accordance with Equation \(\ref{3.5.10}\) \[\psi _{n}(x)=A\, \sin\dfrac{n\pi x}{L} \label{3.5.14} \]In quantum mechanics, the particle in a box model (also known as the infinite potential well or the infinite square well) describes the movement of a free particle in a small space surrounded by impenetrable barriers. The model is mainly used as a hypothetical example to illustrate the differences between classical and quantum systems. In classical systems, for example, a particle trapped inside a large box can move at any speed within the box and it is no more likely to be fo. A particle in a 2-dimensional box is a fundamental quantum mechanical approximation describing the translational motion of a single particle confined inside an infinitely deep well from which it .The particle in a box model lets us consider a simple version of the Schrödinger equation. Before we simplify, let's take another look at the full Hamiltonian for a particle-wave in three dimensions (see equation 2.2.2) and the simplest form .
Explain why the energy of a quantum particle in a box is quantized. Describe the physical meaning of stationary solutions to Schrӧdinger’s equation and the connection of these solutions with time-dependent quantum states. Explain .The Particle in a Box (PIB) is a simple model that helps illustrate the behavior of electrons confined within atoms and molecules. It serves as a useful tool to introduce key quantum concepts: Energetic Quantization: Energy levels in .
• Particle in a box approximation – you solve the Schrödinger equation. Practice Questions 1. The energy levels of the particle in a box are given by εn = ℏ2n2p2/2 mL 2. (a) Why does the .Approximately, therefore the particle in a box in a finite potential energy well can be considered as a first (crude) approximation model of an atom. At least for the purpose of demonstrating the .
exercícios programação fresa cnc resolvidos
We will show how this relationship can be derived from the results of the 1D particle in a box. Additionally, we will show how the particle in a box model can be applied to make .
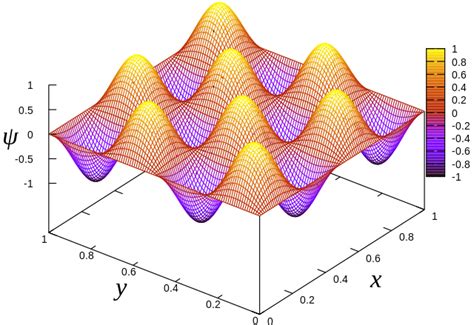
Free particle and the particle in a box. Schrödinger equation is a 2nd-order diff. eq. 2 ∂2ψ ( x ) − + V ( x )ψ ( x Eψ ( x. ) 2m ∂x2. We can find two independent solutions φ. ( x ) and φ. CONCEPT:. Energy Levels in a 1D Particle-in-a-Box System and Variational Method. For a particle confined in a 1D box (potential energy ( V = 0 ) inside the box and ( \(V = \infty \)) outside), the energy levels are quantized, with the ground state energy denoted as ( E 0). The expectation value of the Hamiltonian with a trial wave function provides an estimate of the .Step 1: Define the Potential Energy V. The potential energy is 0 inside the box (V=0 for 0L). We assume the walls have infinite potential energy to ensure that the particle .
Why in the particle in a box model do the values of n begin at 1 but in the harmonic oscillator they begin at 0? I understand what the wave-functions and their corresponding probabilities look like and that the PIB has 0 nodes for n=1 which means the number of nodes is n-1, so for n = 0 it would have -1 nodes which is physically unreasonable.
Free particle and the particle in a box . The energy spacings increase as the box size decreases. 1 E ∝ a2. 5.61 Fall 2007 Lecture #7 page 5 We’ve solved some simple quantum mechanics problems! The P-I-B model is a good approximation for . The Particle-In-A-Box approximation. Electrons in the \( \pi \)-electron system of a conjugated aromatic compound are not restricted to specific nuclei but are free to move throughout the system. In a linear conjugated system the potential energy of the electrons will vary along the chain, being lowest near the nuclei and highest between them.Figure \(\PageIndex{2}\): Visualizing the first six wavefunctions and associated probability densities for a particle in a two-dimensional square box (\(L_x=L_y=L\)).Use the slide bar to independently change either \(n_x\) or \(n_y\) quantum number and see the changing wavefunction. Unlike in the one-dimensional analoge, where nodes in the wavefunction are .
It states that we cannot know both the position and momemtum of a quantum particle with complete certainty. We will show how this relationship can be derived from the results of the 1D particle in a box. Additionally, we will show how the particle in a box model can be applied to make predictions on real systems. 4.5.5.2. Learning Goals:#Classical vs Quantum particle in a box#. Particle in a box is a toy model of electron (or atom, molecule, small quantum object) trapped in some region of space \([0,L]\).. The positional information of a quantum “particle” is described by a quantum wave function \(\psi(x)\) which is obtained by solving Schrödinger equation with boundary conditions .9 Particle-in-a-box(PIB) 1. Consider a linear poly-ene. 2. Theelectrons are completelydelocalized insidethepoly-ene, butcannotleavethe molecular framework. 3. Let us approximate this system by a one-dimensional box, of length L. The potential energy of the electrons inside the polyenes can be approximated by the figure below. 4.
The conventional quantum mechanical problem of a particle in a one-dimensional box can be made more interesting chemically without introducing any more difficult mathematics by employing a potential diagram rather than the usual infinite well.
Answer to 17. The molecular orbital energies of butadiene. 17. The molecular orbital energies of butadiene CH2=CH-CH=CH2 can be ap- proximately represented either using the particle-in-a-box model or using the Hückel approximation (although the latter approximation significantly under- estimates the energies of anti-bonding molecular orbitals).The particle-in-a-box model for motion in one or two dimensions discussed earlier can obviously be extended to three dimensions. . This same spherical box model has also been used to describe the valence electrons in quasi-spherical nano-clusters of metal atoms such as \(Cs_n\), \(Cu_n\), \(Na_n\), \(Au_n\), \(Ag_n\), and their positive and .
and sketch this function: the particle is more likely to be found in the left-hand half of the box. Now, suppose the time is t = 4 m L 2 / h, so e − i π h t 4 m L 2 = − 1. At this time, ψ (x, 2 L 2 / h) = 1 L (− sin π x L + sin 2 π x L) and it’s easy to see that .An electron moving in a thin metal wire is a reasonable approximation of a particle in a one-dimensional infinite well. The potential inside the wire is constant on average but rises at each end. Suppose the electron is in a wire 1.0 cm long. (a) Compute the ground-state energy for the electron. (b) If the electron
If you consider your wave-function, with sinusoidal changes within a box, the particle is considered a wave with energies in a probable range. So of course the attributes of a wave will change given the length of your box. The choice of boundary condition affects the physical probabilities. An electron moving in a thin metal wire is a reasonable approximation of a particle in a one-dimensional infinite well. The potential inside the wire is constant on average but rises sharply at each end. Suppose the electron is in a wire 1.0 cm long. 1) Find the ground-state energy for the electron.
particle in a box wikipedia
Answer to Based on the particle in a one-dimensional box. Science; Chemistry; Chemistry questions and answers; Based on the particle in a one-dimensional box approximation for polyenes, suggest where along the line segment the n=1 to n=2 electronic transiton would most likely take place. explain your choiceAlthough a particle in such potential is an idealization, it is a very important problem because: a) Exact solutions are obtained from the Schrodinger Equation. b) It demonstrates important features of quantum-mechanical problems. c) This potential is a good approximation to some real situations such as a free electron in a metal. Question: To a crude first approximation a pi electron in a linear polyene may be considered as a particle in a 1D box. To a crude first approximation a pi electron in a linear polyene may be considered as a particle in a 1 D box. Here’s the best way to solve it. Solution. The quantum particle in the 1D box problem can be expanded to consider a particle within a higher dimensions as demonstrated elsewhere for a quantum particle in a 2D box.Here we continue the expansion into a particle trapped in a 3D box with three lengths \(L_x\), \(L_y\), and \(L_z\). As with the other systems, there is NO FORCE (i.e., no potential) acting on .
exchange 2016 disable distribution email box
where the momentum = ℏ and the numbers n x, n y, and n z are any set of integers– positive, negative, or zero. We can imagine setting up coordinate axes k x, k y, and k z and putting a point everywhere in k-space that there is an allowed state. Notice that as the size of the box gets bigger, the states get closer together in k-space. Since E = ℏ 2 k 2 ∕ 2 m, the lower energy .
We will introduce the Drude model, a reasonable description of electrons in metals. The quasi-static approximation will then let us take into account the nanoscale size of the particles. Additionally, the generalization of the Clausius–Mossotti relation will let us consider a particle embedded in a dielectric medium and the effect of its .The density of states related to volume V and N countable energy levels is defined as: = = (()). Because the smallest allowed change of momentum for a particle in a box of dimension and length is () = (/), the volume-related density of states for continuous energy levels is obtained in the limit as ():= (()), Here, is the spatial dimension of the considered system and the wave vector. The particle in a box is the very first example most people see of a bound state problem. These are a class of quantum mechanical problems whereby we see, by simple mathematics, that the energy levels of certain quantum systems are discretely quantized. . Using Sterling's approximation, $\log{N!}\sim N\log{N}-N$, we arrive at
particle in a box model
We now turn our attention to a generalization of the 1-dimensional quantum box to 3 dimensions. The 3 - dimensional quantum box is shown schematically in figure 1.5. It extends along 0 ^ x ^ L x, 0 ^ у ^ L y, 0 ^ z ^ L z. Outside this region the potential is infinite so that the wavefunction ¥ is zero at the faces of the box.
Particle in a box with finite-potential walls# 2.6.1. . Consider the case the situation of electrons in a metal. We saw when looking at the Photoelectric effect that a reasonable approximation of the potential that confines the electrons within the metal had a finite depth. This is the potential we will now consider and it is be far the most .
As with Example 7.4.1 , we recognize that unperturbed component of the problem (Equation \(\ref{7.4.2}\)) is the particle in an infinitely high well. For this system, the unperturbed Hamiltonian and solution is the particle in an infinitely high box and the perturbation is a shift of the potential within half a box by \(V_o\).
particle in a box function
Nearly all CNC machine uses a Cartesian coordinate system based on an X, Y, and Z-axis. This system allows a machine to move in a specific direction along a specific plane. Reduce the Cartesian system to its basics, and you have a familiar number line.
particle in a box approximation of metals|particle in a box wikipedia